Shares of Biogen BIIB rose 2% on Monday after the company announced that the FDA approved a higher dose of its blockbuster spinal muscular atrophy (SMA) drug Spinraza.
The new dosing regimen simplifies administration by offering two initial 50 mg doses spaced 14 days apart, followed by a 28 mg maintenance dose every four months. This approach reduces the dosing frequency compared to the low-dose version, which requires four loading doses of 12mg administered at an interval of 14 days, followed by a 12 mg maintenance dose every four months.
The FDA approval is supported by data from the phase II/III DEVOTE study, which achieved its primary endpoint of statistically significant improvement in motor function in infants with SMA treated with a higher dose of Spinraza. The drug secured approval for a similar indication in the EU in January, based on this data.
Per Biogen, a higher dose regimen of the drug has the potential to slow neurodegeneration faster through greater reductions in neurofilament compared to the low-dose version. Patients who transitioned to the higher dose regimen from the low-dose version experienced improvements in motor function after the move.
Biogen expects to launch the high-dose regimen in the coming weeks.
BIIB Stock Performance
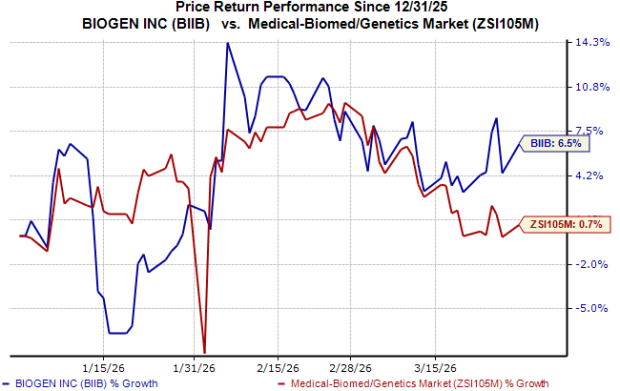
Shares of Biogen have outperformed the industry year to date, as seen in the chart below.
Image Source: Zacks Investment Research
Spinraza: A Key Revenue Driver for Biogen
The drug was discovered and initially developed by Ionis Pharmaceuticals IONS. Post approval for the SMA indication, Biogen in-licensed the drug’s worldwide rights from Ionis and assumed responsibility for its future development, manufacturing and commercialization.
Biogen’s Spinraza faces competition from Novartis’ NVS gene therapy Zolgensma and Roche’s RHHBY oral drug Evrysdi, both of which are approved for the SMA indication.
Competition from both the NVS and RHHBY therapies has shifted market dynamics, negatively impacting Spinraza’s sales in the United States. Due to increased competitive pressure, sales of the Biogen drug have been declining in the past few years. Spinraza sales declined almost 2% year over year in 2025 to $1.55 billion.
We expect this latest approval of Spinraza to help Biogen address competitive concerns and drive growth in future quarters.
Biogen Inc. Price
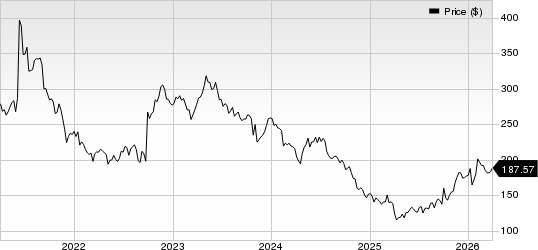
Biogen Inc. price | Biogen Inc. Quote
BIIB’s Zacks Rank
Biogen currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Zacks' Research Chief Names "Stock Most Likely to Double"
Our team of experts has just released the 5 stocks with the greatest probability of gaining +100% or more in the coming months. Of those 5, Director of Research Sheraz Mian highlights the one stock set to climb highest.
This top pick is a little-known satellite-based communications firm. Space is projected to become a trillion dollar industry, and this company's customer base is growing fast. Analysts have forecasted a major revenue breakout in 2025. Of course, all our elite picks aren't winners but this one could far surpass earlier Zacks' Stocks Set to Double like Hims & Hers Health, which shot up +209%.
Free: See Our Top Stock And 4 Runners UpWant the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Novartis AG (NVS): Free Stock Analysis Report
Biogen Inc. (BIIB): Free Stock Analysis Report
Roche Holding AG (RHHBY): Free Stock Analysis Report
Ionis Pharmaceuticals, Inc. (IONS): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).