We expect investors to focus on pipeline updates when Keros Therapeutics KROS reports fourth-quarter results shortly. The Zacks Consensus Estimate for sales and loss per share is pegged at $6.50 million and 49 cents, respectively.
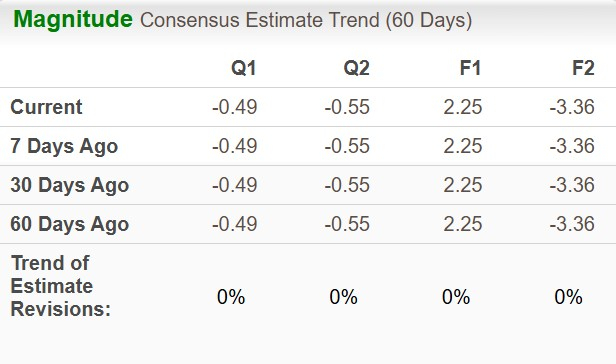
Earnings estimate for 2025 has remained stable over the past 60 days, while loss per share estimate for 2026 has also remained unchanged in the same time frame.
Image Source: Zacks Investment Research
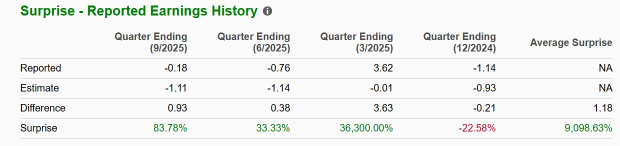
KROS’ Earnings Surprise History
KROS has a good track record. The company beat earnings estimates in three of the trailing four quarters and missed in the remaining one, delivering an average surprise of 9,098.63%. In the last reported quarter, the company beat earnings estimates by 83.78%.
Image Source: Zacks Investment Research
What Our Model Predicts for KROS
Per our proven model, the combination of a positive Earnings ESP and a Zacks Rank #1 (Strong Buy), 2 (Buy) or 3 (Hold) increases the odds of an earnings beat.
Earnings ESP for KROS is 0.00%. The company currently carries a Zacks Rank #3. You can uncover the best stocks to buy or sell before they're reported with our Earnings ESP Filter.
Focus on KROS’ Pipeline Progress
Since Keros is a clinical-stage biopharmaceutical company, it does not generate any product sales. The top line comprises license and service revenues that it receives under its collaboration with Takeda Pharmaceuticals TAK.
In such a scenario, investors’ focus will be primarily on pipeline progress when the company reports quarterly results.
KROS is focused on developing and commercializing novel therapeutics to treat a wide range of patients with disorders that are linked to dysfunctional signaling of the transforming growth factor-beta, or TGF-ß, family of proteins.
The recent pipeline progress has been encouraging. Lead product candidate, rinvatercept (KER-065), is designed to bind to and inhibit TGF-ß ligands, including myostatin (GDF8) and activin A, which are negative regulators of muscle and bone mass and strength. The company is advancing rinvatercept for the treatment of neuromuscular disorders, initially targeting Duchenne muscular dystrophy (DMD).
In March 2025, Keros reported initial top-line results from a phase I study of rinvatercept in healthy volunteers. The FDA granted orphan drug designation for rinvatercept to treat DMD.
The company plans to begin a phase II trial in patients with DMD in the first quarter of 2026. Further updates on the same are expected with the upcoming results.
KROS plans to engage with the regulators on the design of a phase II study evaluating rinvatercept in patients with amyotrophic lateral sclerosis (ALS) in the second half of 2026.
In December 2024, Keros Therapeutics entered into an exclusive license agreement with Takeda Pharmaceuticals (TAK) to develop, manufacture, and commercialize its another pipeline candidate, elritercept, worldwide, excluding mainland China, Hong Kong and Macau. The agreement, which turned effective on Jan. 16, 2025, provides Keros with funds in the form of milestone payments.
A phase III study, RENEW, is evaluating elritercept for the treatment of anemia and thrombocytopenia in patients with very low-, low-, or intermediate-risk myelodysplastic syndromes (MDS).
In July 2025, the company announced that the first patient had been dosed in the RENEW study, triggering a $10 million milestone payment to Keros under its global licensing agreement with Takeda.
Another phase II study, RESTORE, is evaluating elritercept in patients with myelofibrosis-associated cytopenias.
Meanwhile, Keros has taken deliberate steps to sharpen its strategic focus and strengthen capital efficiency. The company was previously advancing cibotercept for pulmonary arterial hypertension, but in August 2025, it discontinued the program and redirected resources toward its lead asset, KER-065, which currently appears to offer more compelling potential.
To support this realignment, management implemented a workforce reduction of approximately 45%, resulting in a streamlined organization of about 85 full-time employees. These actions are expected to generate average annualized cost savings of roughly $17 million. In August 2025, Keros announced board and leadership changes, which are aimed at reinforcing a leaner operating structure and tighter strategic execution.
Hence, operating expenses have likely declined in the fourth quarter.
KROS’ Price Performance and Valuation
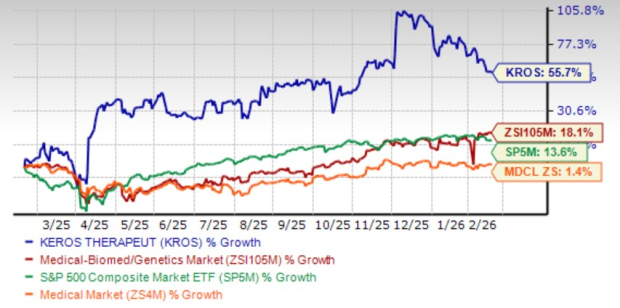
Shares of the company have surged 55.7% in the past year compared with the industry’s gain of 18.1%. The stock has also outperformed the sector and the S&P 500 Index.
Image Source: Zacks Investment Research
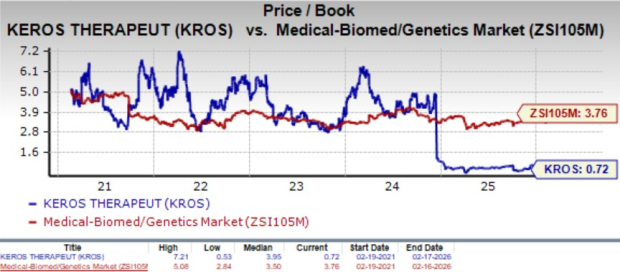
Going by the price/book ratio, KROS is quite inexpensive. Shares currently trade at 0.72x tangible book value, lower than the industry’s average of 3.76X and its mean of 3.95X.
Image Source: Zacks Investment Research
Stay Invested in KROS Stock
Investors in clinical-stage companies primarily base their decisions on the strength of the pipeline and its long-term potential. DMD is a highly competitive and scientifically complex market.
Sarepta Therapeutics SRPT is a formidable player in this space, with a strong DMD franchise that includes exon-skipping therapies, such as Exondys 51, Vyondys 53 and Amondys 45. In June 2023, Sarepta obtained accelerated approval from the FDA for Elevidys, an adeno-associated virus-based gene therapy for the treatment of ambulatory pediatric patients aged four through five years with DMD who have a confirmed mutation in the DMD gene.
In June 2024, the FDA granted full approval to Elevidys for the treatment of ambulatory individuals aged four years and older, along with accelerated approval for the treatment of non-ambulatory individuals aged four years and older.
Against this backdrop, the successful development and eventual commercialization of its pipeline candidates are critical for Keros Therapeutics. The stock has delivered strong gains over the past year, which could limit near-term upside.
Prospective investors may prefer to wait for more attractive entry points, while existing shareholders could consider maintaining positions.
The company has made meaningful progress with its lead DMD program, and any positive clinical or regulatory development for KER-065 could act as a significant catalyst and support further upside in KROS shares.
You can see the complete list of today’s Zacks #1 Rank stocks here.
5 Stocks Set to Double
Each was handpicked by a Zacks expert as the #1 favorite stock to gain +100% or more in the coming year. While not all picks can be winners, previous recommendations have soared +112%, +171%, +209% and +232%.
Most of the stocks in this report are flying under Wall Street radar, which provides a great opportunity to get in on the ground floor.
Today, See These 5 Potential Home Runs >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Sarepta Therapeutics, Inc. (SRPT): Free Stock Analysis Report
Takeda Pharmaceutical Co. (TAK): Free Stock Analysis Report
Keros Therapeutics, Inc. (KROS): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).