Rocket Pharmaceuticals RCKT announced that the FDA has granted accelerated approval to its gene therapy Kresladi (marnetegragene autotemcel) to treat patients with severe leukocyte adhesion deficiency-I (LAD-I), an ultra-rare genetic disorder.
Following the latest nod, Kresladi became the first gene therapy to be approved by the FDA for treating children with severe LAD-I due to biallelic variants in ITGB2 without an available human leukocyte antigen-matched sibling donor for allogeneic hematopoietic stem cell transplant.
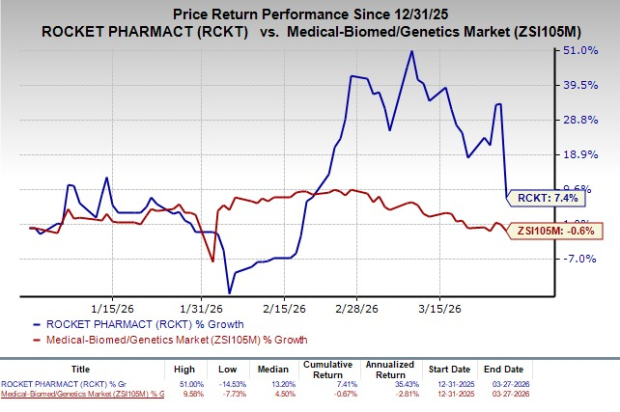
Despite the FDA nod, shares of Rocket Pharmaceuticals were down 20% on Friday following the announcement of the news.
The stock has risen 7.4% so far this year against the industry’s decline of 0.6%.
Image Source: Zacks Investment Research
More on the FDA Nod for RCKT’s Kresladi
Last October, the FDA accepted RCKT’s resubmitted biologics license application (BLA) for Kresladi to treat patients with severe LAD-I. A final decision was due on March 28, 2026.
The FDA nod for Kresladi came a day before its actual timeline. Per the company, this treatment was approved early because it increased specific markers (CD18 and CD11a) on immune cells. Its actual clinical benefits will be confirmed through using long-term study results and post-marketing data collection.
This might have hurt investor sentiments and resulted in the stock declining on Friday.
With the FDA approval for Kresladi, the company received a Rare Pediatric Disease Priority Review Voucher (PRV), which is an incentive given by the FDA to encourage the development of drugs and biologics for rare and serious diseases.
Rocket Pharmaceuticals plans to explore strategic options to monetize PRV to improve its financial flexibility while increasing shareholder value.
LAD-I is a rare, severe pediatric genetic disorder that is caused by a defect in the ITGB2 gene. This leads to a deficiency in CD18 and can cause recurrent and life-threatening infections, fatally affecting the immune system. The disease has high early childhood mortality without treatment.
RCKT’s Kresladi Approval Comes After Hurdle
We remind investors that in June 2024, the FDA issued a complete response letter (CRL) to the BLA for Kresladi for treating LAD-I.
Per the CRL, the FDA sought "limited" additional information on the Chemistry Manufacturing and Controls (CMC), which was part of the BLA submission.
Notably, this was the second time that the FDA had requested additional information from the company on Kresladi’s CMC. In February 2024, the FDA extended the review period of the Kresladi BLA by an additional three months.
Now that the FDA has approved Kresladi, it is likely to give RCKT its first commercial product and should provide a steady source of revenue.
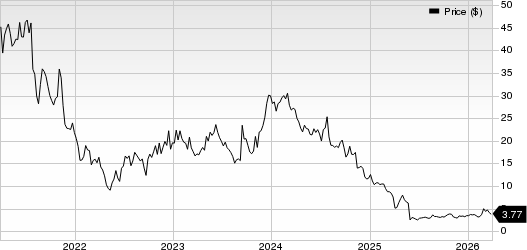
Rocket Pharmaceuticals, Inc. Price
Rocket Pharmaceuticals, Inc. price | Rocket Pharmaceuticals, Inc. Quote
RCKT's Zacks Rank & Stocks to Consider
Rocket Pharmaceuticals currently carries a Zacks Rank #3 (Hold).
Some better-ranked stocks in the biotech sector are Prothena PRTA, ANI Pharmaceuticals ANIP and Replimune Group REPL, each carrying a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Over the past 60 days, estimates for Prothena’s 2026 earnings per share have increased from 20 cents to 36 cents, while loss per share estimates for 2027 have narrowed from $1.20 to 98 cents. PRTA’s shares have decreased 3.1% year to date.
Prothena’s earnings beat estimates in one of the trailing four quarters and missed on the remaining three occasions, with the average negative surprise being 24.69%.
Over the past 60 days, estimates for ANI Pharmaceuticals’ 2026 earnings per share have increased from $8.28 to $8.99, while the same for 2027 have increased from $9.25 to $10.10. ANIP’s shares have fallen 7.7% year to date.
ANI Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 22.21%.
Over the past 60 days, estimates for Replimune’s 2026 loss per share have narrowed from $3.61 to $3.40, while the same for 2027 have narrowed from $2.58 to $2.46. REPL’s shares have tanked 25.8% year to date.
Replimune’s earnings beat estimates in one of the trailing four quarters and missed on the remaining three occasions, with the average negative surprise being 4.46%.
Free Report: Profiting from the 2nd Wave of AI Explosion
The next phase of the AI explosion is poised to create significant wealth for investors, especially those who get in early. It will add literally trillion of dollars to the economy and revolutionize nearly every part of our lives.
Investors who bought shares like Nvidia at the right time have had a shot at huge gains.
But the rocket ride in the "first wave" of AI stocks may soon come to an end. The sharp upward trajectory of these stocks will begin to level off, leaving exponential growth to a new wave of cutting-edge companies.
Zacks' AI Boom 2.0: The Second Wave report reveals 4 under-the-radar companies that may soon be shining stars of AI’s next leap forward.
Access AI Boom 2.0 now, absolutely free >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Prothena Corporation plc (PRTA): Free Stock Analysis Report
ANI Pharmaceuticals, Inc. (ANIP): Free Stock Analysis Report
Rocket Pharmaceuticals, Inc. (RCKT): Free Stock Analysis Report
Replimune Group, Inc. (REPL): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).