Nkarta NKTX shares surged 15.7% on Thursday, reflecting strong investor optimism following a meaningful regulatory update tied to its lead autoimmune program, NKX019, an investigational CAR-NK cell therapy. The rally was observed after NKTX reached an agreement with the FDA on key protocol changes that could materially improve study efficiency, patient accessibility and the therapy’s broader commercial potential.
Per Nkarta, the FDA has cleared amendments to its ongoing Ntrust-1 and Ntrust-2 studies, most notably enabling outpatient dosing of NKX019. This change significantly reduces monitoring requirements from 24 hours to just two, eliminating the need for overnight hospital stays. Management believes that such a shift could lower treatment burden, improve patient enrollment rates and expand administration into community rheumatology centers, thereby accelerating study execution and future scalability.
The FDA agreement also introduces greater clinical flexibility. Nkarta now has the option to re-dose patients in both studies, a move that could enhance therapeutic durability and optimize individual patient outcomes. In addition, the planned inclusion of a rheumatoid arthritis cohort in the Ntrust-2 study broadens NKX019’s addressable market into a high-prevalence autoimmune indication with high unmet need, signaling potential for significant pipeline expansion.
Investors also appear encouraged by upcoming catalysts, with initial clinical data from these studies expected to be presented at a medical meeting later this year. Taken together, the combination of reduced logistical barriers, expanded study scope and near-term data visibility likely underpinned the sharp upward move in Nkarta stock.
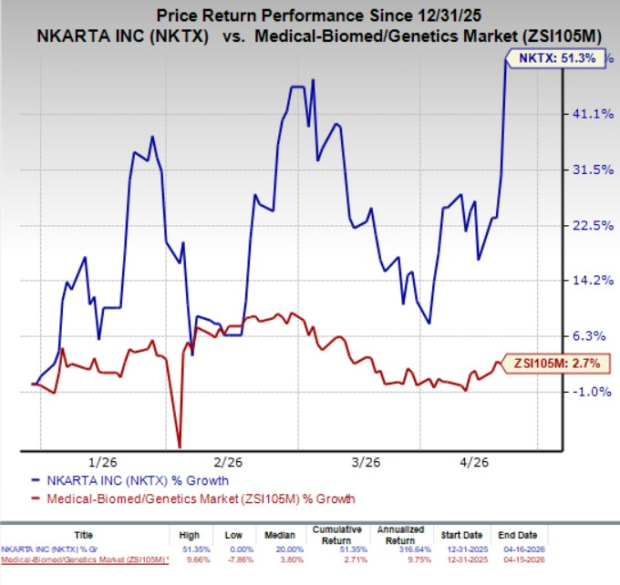
Year to date, NKTX stock has surged 51.3% compared with the industry’s 2.7% growth.
Image Source: Zacks Investment Research
More on NKTX’s NKX019 Studies for Autoimmune Diseases
Nkarta’s Ntrust-1 and Ntrust-2 are dose-escalation clinical studies designed to build on prior academic evidence showing durable, drug-free remissions in autoimmune diseases following CD19-targeted cell therapies. Both ongoing studies are primarily evaluating the safety profile of NKX019 while also assessing its potential to induce sustained remission via a “reset” of the immune system through the elimination of pathogenic B cells.
The studies are currently enrolling patients across a range of autoimmune indications, including lupus nephritis, primary membranous nephropathy, systemic sclerosis, idiopathic inflammatory myopathy, and ANCA-associated vasculitis. Rheumatoid arthritis is expected to be added as an additional cohort once the amended protocol receives final clearance, which would further expand the therapy’s target population.
Per the study design, patients receive a three-dose cycle of NKX019 administered on Days 0, 3 and 7, following lymphodepletion with fludarabine and cyclophosphamide or cyclophosphamide alone, if they have significant cytopenia at baseline. Notably, no supplemental cytokines or antibody-based therapies are used, allowing researchers to evaluate NKX019 as a single-agent treatment and potentially streamline its regulatory pathway. Patients may also receive additional treatment cycles, if needed, to deepen or restore therapeutic response.
Nkarta, Inc. Price and Consensus
Nkarta, Inc. price-consensus-chart | Nkarta, Inc. Quote
NKTX’s Zacks Rank and Stocks to Consider
Nkarta currently carries a Zacks Rank #4 (Sell).
Some better-ranked stocks in the biotech sector are Catalyst Pharmaceuticals CPRX, ADMA Biologics ADMA and Inovio Pharmaceuticals INO. While CPRX and ADMA sport a Zacks Rank #1 (Strong Buy) each, INO carries a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.
Over the past 60 days, estimates for Catalyst Pharmaceuticals’ 2026 EPS have increased from $2.55 to $2.87. CPRX shares have gained 11% year to date.
Catalyst Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 35.19%.
Over the past 60 days, estimates for ADMA Biologics’ 2026 EPS have increased from 85 cents to 93 cents. ADMA shares have plummeted 41.2% year to date.
ADMA Biologics’ earnings beat estimates in one of the trailing three quarters, matched once and missed on the remaining occasion, with the average negative surprise being 1.79%.
Over the past 60 days, estimates for Inovio Pharmaceuticals’ 2026 loss per share have narrowed from $1.26 to $1.06. INO shares have plunged 33.4% year to date.
Inovio Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 57.94%.
#1 Semiconductor Stock to Buy (Not NVDA)
The incredible demand for data is fueling the market's next digital gold rush. As data centers continue to be built and constantly upgraded, the companies that provide the hardware for these behemoths will become the NVIDIAs of tomorrow.
One under-the-radar chipmaker is uniquely positioned to take advantage of the next growth stage of this market. It specializes in semiconductor products that titans like NVIDIA don't build. It's just beginning to enter the spotlight, which is exactly where you want to be.
See This Stock Now for Free >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Catalyst Pharmaceuticals, Inc. (CPRX): Free Stock Analysis Report
Inovio Pharmaceuticals, Inc. (INO): Free Stock Analysis Report
ADMA Biologics Inc (ADMA): Free Stock Analysis Report
Nkarta, Inc. (NKTX): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).