Kymera Therapeutics KYMR announced that Gilead Sciences GILD has exercised its option to obtain an exclusive license for Kymera’s KT-200, a first-in-class, oral cyclin-dependent kinase 2 (CDK2) molecular glue degrader (MGD) under their strategic collaboration agreement.
Concurrently, Kymera is entitled to a $45 million milestone payment.
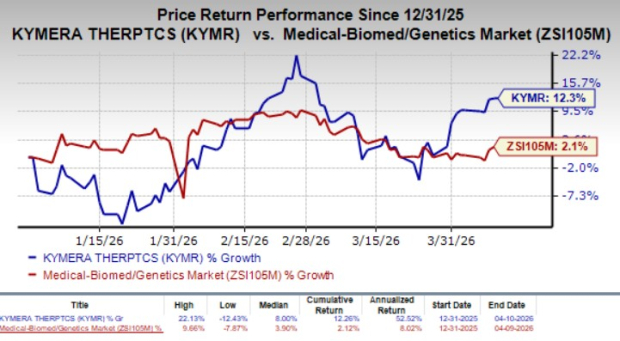
Year to date, the shares of KYMR have rallied 12.3% compared with the industry’s 2.1% growth.
Image Source: Zacks Investment Research
KYMR-GILD Agreement for Molecular Glue CDK2 Degraders
In June 2025, Kymera entered into an exclusive option and license agreement with Gilead to accelerate the development and commercialization of a novel MGD program targeting CDK2 with broad oncology treatment potential, in breast cancer and other solid tumors.
Per the agreement, Kymera is eligible to receive up to $750 million in total payments contingent on the achievement of specified development, regulatory and commercial milestones. The company has already realized $85 million in upfront and option exercise payments. Kymera stands to benefit from tiered royalties on net product sales, ranging from the high single digits to the mid-teens.
While Kymera leads all research activities for the CDK2 program, Gilead retains global rights to develop, manufacture and commercialize any products arising from the collaboration.
CDK2-directed MGDs are an emerging class of therapies that selectively eliminate the CDK2 protein — a key driver of tumor growth — rather than merely inhibiting its activity. This targeted approach has the potential to deliver safer and more effective oral treatments for CDK2-driven cancers, potentially improving outcomes, particularly in advanced breast cancer, where treatment options remain limited.
KT-200 demonstrated low-nanomolar CDK2 degradation, strong activity in CCNE1-driven models, potential brain penetration and a favorable safety profile in preclinical studies. It would be Kymera’s first molecular glue candidate to advance into clinical development.
Gilead plans to advance KT-200 into investigational new drug (IND)-enabling studies, with the goal of supporting an IND filing in 2027.
Kymera’s Zacks Rank & Stocks to Consider
KYMR currently carries a Zacks Rank #3 (Hold).
Some top-ranked stocks in the biotech sector are Catalyst Pharmaceuticals CPRX and Indivior Pharmaceuticals INDV, each currently sporting a Zacks Rank #1 (Strong Buy). You can see the complete list of today’s Zacks #1 Rank stocks here.
Over the past 60 days, estimates for Catalyst Pharmaceuticals’ 2026 earnings per share have risen from $2.55 to $2.87. CPRX shares have gained 9.68% year to date.
Catalyst Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 35.19%.
Over the past 60 days, estimates for Indivior Pharmaceuticals’ 2026 earnings per share have risen from $2.89 to $3.08. INDV shares have lost 9.95% year to date.
Indivior Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 74.53%.
Zacks Names #1 Semiconductor Stock
This under-the-radar company specializes in semiconductor products that titans like NVIDIA don't build. It's uniquely positioned to take advantage of the next growth stage of this market. And it's just beginning to enter the spotlight, which is exactly where you want to be.
With strong earnings growth and an expanding customer base, it's positioned to feed the rampant demand for Artificial Intelligence, Machine Learning, and Internet of Things. Global semiconductor manufacturing is projected to explode from $452 billion in 2021 to $971 billion by 2028.
See This Stock Now for Free >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Gilead Sciences, Inc. (GILD): Free Stock Analysis Report
Catalyst Pharmaceuticals, Inc. (CPRX): Free Stock Analysis Report
Kymera Therapeutics, Inc. (KYMR): Free Stock Analysis Report
Indivior Pharmaceuticals Inc. (INDV): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).