Travere Therapeutics TVTX shares surged 37.2% on Tuesday after the company announced that the FDA approved Filspari (sparsentan) for a second rare kidney disease. The approval has expanded the drug’s use to reduce proteinuria in adult and pediatric patients (aged 8 years and older) with focal segmental glomerulosclerosis (FSGS) without nephrotic syndrome, making it the first and only medicine approved by the FDA for this indication.
Filspari was earlier approved to slow kidney function decline in adults with primary IgA nephropathy (IgAN) who are at risk of disease progression. Per TVTX, it is currently the most commonly prescribed FDA-approved medicine for this indication.
Patients with FSGS who do not present with nephrotic syndrome encompass multiple subtypes of the disease and represent a population consistent with KDIGO guidelines for the management of glomerular disorders. Nephrotic syndrome is typically defined by the presence of three concurrent criteria — proteinuria exceeding 3.5 g/24h, edema and serum albumin levels below 3.0 g/dL. The label aligns with the KDIGO clinical practice guidelines for treating patients with FSGS.
Rationale Behind the FDA Nod for TVTX’s Filspari for FSGS
Travere Therapeutics’ Filspari regulatory filing was primarily based on robust clinical evidence from the largest head-to-head interventional study in FSGS to date, the phase III DUPLEX study. In the study, treatment with the drug resulted in rapid, superior and sustained reductions in proteinuria compared with the maximum labeled dose of Sanofi’s Avapro (irbesartan) across adult and pediatric patients.
Results from the DUPLEX study demonstrated that patients treated with Filspari in the overall study population experienced a statistically significant 46% reduction in proteinuria from baseline to week 108 compared to 30% for those treated with Avapro. Among patients without nephrotic syndrome, Filspari outperformed Avapro on both proteinuria reduction and eGFR measures. The drug delivered a 48% reduction in proteinuria from baseline to week 108 compared to a 27% decline with Avapro, a statistically significant difference. Filspari-treated patients without nephrotic syndrome also showed a modest advantage in preserving kidney function, with a treatment difference in eGFR of 1.1 mL/min/1.73 m² based on mean change from baseline over the same period (–11.3 vs. –12.4).
Importantly, the therapy was generally well tolerated across both adult and pediatric populations, with a safety profile comparable to Avapro and consistent with findings from prior clinical programs.
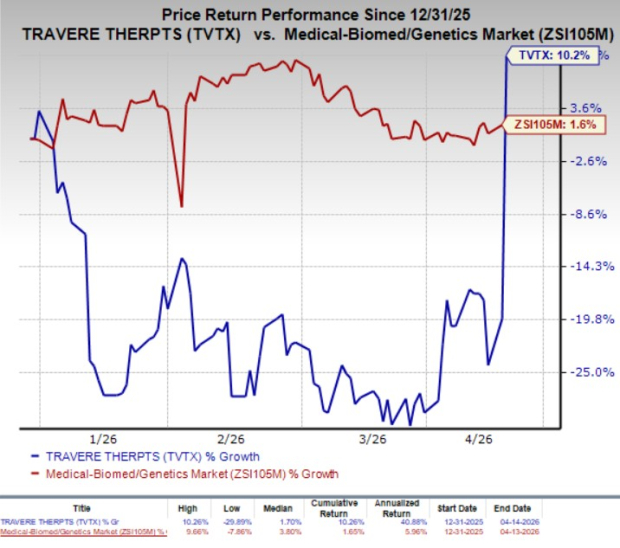
Year to date, Travere Therapeutics stock has gained 10.2% compared with the industry’s 1.6% growth.
Image Source: Zacks Investment Research
FSGS is a rare kidney disorder affecting more than 30,000 patients in the United States. The disease is marked by progressive kidney scarring and proteinuria (protein in the urine), where leaked protein damages kidney structures and drives disease progression. Patients often experience edema, low blood albumin, abnormal lipid levels and hypertension, with many ultimately progressing to kidney failure.
In patients without nephrotic syndrome, FSGS is primarily driven by glomerular stress along with the activation of inflammatory and fibrotic pathways. Filspari is an oral, non-immunosuppressive therapy with a dual mechanism of action that targets these underlying processes by inhibiting endothelin A and angiotensin II receptors, a combination believed to help preserve kidney function and limit disease progression.
The latest approval strengthens Travere Therapeutics’ position in rare kidney diseases and meaningfully expands Filspari’s addressable market to more than 100,000 patients in the United States with FSGS and IgAN who have limited treatment options. Importantly, Filspari is immediately available for prescription, enabling nephrologists to begin treating eligible FSGS patients without delay.
Travere Therapeutics, Inc. Price and Consensus
Travere Therapeutics, Inc. price-consensus-chart | Travere Therapeutics, Inc. Quote
TVTX's Zacks Rank & Stocks to Consider
Travere Therapeutics currently carries a Zacks Rank #3 (Hold).
Some better-ranked stocks in the biotech sector are Catalyst Pharmaceuticals CPRX, ADMA Biologics ADMA and Inovio Pharmaceuticals INO. While CPRX and ADMA sport a Zacks Rank #1 (Strong Buy) each, INO carries a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.
Over the past 60 days, estimates for Catalyst Pharmaceuticals’ 2026 EPS have increased from $2.55 to $2.87. CPRX shares have gained 12.3% year to date.
Catalyst Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 35.19%.
Over the past 60 days, estimates for ADMA Biologics’ 2026 EPS have increased from 85 cents to 96 cents. ADMA shares have plummeted 42.5% year to date.
ADMA Biologics’ earnings beat estimates in one of the trailing three quarters, matched once and missed on the remaining occasion, with the average negative surprise being 1.79%.
Over the past 60 days, estimates for Inovio Pharmaceuticals’ 2026 loss per share have narrowed from $1.26 to $1.06. INO shares have plunged 36.3% year to date.
Inovio Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 57.94%.
5 Stocks Set to Double
Each was handpicked by a Zacks expert as the #1 favorite stock to gain +100% or more in the coming year. While not all picks can be winners, previous recommendations have soared +112%, +171%, +209% and +232%.
Most of the stocks in this report are flying under Wall Street radar, which provides a great opportunity to get in on the ground floor.
Today, See These 5 Potential Home Runs >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Catalyst Pharmaceuticals, Inc. (CPRX): Free Stock Analysis Report
Inovio Pharmaceuticals, Inc. (INO): Free Stock Analysis Report
ADMA Biologics Inc (ADMA): Free Stock Analysis Report
Travere Therapeutics, Inc. (TVTX): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).